 Lide, (ed.), CRC Handbook of Chemistry and Physics 1999-2000 : A Ready-Reference Book of Chemical and Physical Data (CRC Handbook of Chemistry and Physics, CRC Press, Boca Raton, Florida, USA, 79th edition, 1998. I am grateful to Professor Pekka Pyykkö (University of Helsinki, Finland) who provided the nuclear quadrupole moment data in this and the following two references. Where given, data for certain radioactive nuclei are from this reference. Mason in Multinuclear NMR, Plenum Press, New York, USA, 1987. 
I am grateful to Professor Robin Harris (University of Durham, UK) who provided much of the NMR data, which are copyright 1996 IUPAC, adapted from his contribution contained within this reference. 5, John Wiley & Sons, Chichester, UK, 1996. Harris in Encyclopedia of Nuclear Magnetic Resonance, D.M. Properties of a copper atom This article discussed in detail how to easily find the number of protons, neutrons, and electrons in a copper atom. The number of neutrons depends on the isotope of the element. Magnetogyric ratio, γ (10 7 rad T ‑1 s -1) Copper-63 isotope is used for neutron spectrum measurement Copper-63 isotope is used for studies of high-temperature superconductivity Copper-63 isotope is. Therefore, a copper atom has thirty-five neutrons. Table of NMR-active nucleus propeties of copper Both isotopes have the same properties and are both stable. Both Cu-63 and Cu-65 atoms have 29 protons, but Cu-63 has 34 neutrons and Cu-65 has 36 neutrons. Ĭommon reference compound: ClO 4 in MeCN. How many isotopes does copper have For example, copper has two forms: copper 63, which is 70 of all copper in nature, and copper 65, which makes up 30 of the rest. the argon isotope with 22 neutrons Write isotopic symbols in the form 2X for each isotope. Kuchitsu in Quantities, Units and Symbols in Physical Chemistry, Blackwell Scientific Publications, Oxford, UK, 1988. the potassium isotope with 21 neutrons d. Masses, nuclear spins, and magnetic moments: I.For further information about radioisotopes see Jonghwa Chang's (Korea Atomic Energy Research Institute) Table of the Nuclides.Naturally occurring isotope abundances: Commission on Atomic Weights and Isotopic Abundances report for the International Union of Pure and Applied Chemistry in Isotopic Compositions of the Elements 1989, Pure and Applied Chemistry, 1998, 70, 217.Its discoverer and discovery date are unknown. This table gives information about some radiosotopes of copper, their masses, their half-lives, their modes of decay, their nuclear spins, and their nuclear magnetic moments. 63.55 The chemical element copper is classed as a transition metal. 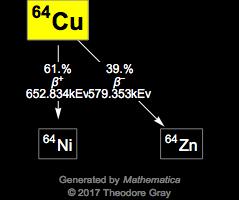
Further data for naturally occuring isotopes of copper are listed above. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
